
It provided conclusive evidence for the existence of nuclei within atoms, leading to the development of the Rutherford atomic model. In summary, Rutherford’s gold foil experiment was a pivotal moment in the history of science. His experiment opened up new avenues of research and inquiry into the nature of matter and energy, influencing generations of scientists and shaping the trajectory of scientific discovery. By demonstrating that the atom is mostly empty space with a concentrated nucleus, Rutherford laid the foundation for modern atomic theory. The gold foil experiment not only paved the way for the development of the Rutherford atomic model but also fundamentally changed our perception of the atom. This revolutionized our understanding of atomic structure and set the stage for further advancements in the field of nuclear physics and chemistry. Rutherford’s atomic model described the atom as having a small, dense, positively charged nucleus surrounded by negatively charged electrons in orbit. The gold foil experiment provided compelling evidence for the existence of a dense nucleus within the atom and shattered the notion that the atom was a solid structure. It was almost as incredible as if you fired a 15-inch shell at a piece of tissue paper, and it came back and hit you.” – Ernest Rutherford “It was quite the most incredible event that has ever happened to me in my life. This astonishing result challenged the prevailing understanding of atomic structure and led Rutherford to propose his now famous atomic model, known as the Rutherford atomic model or the nuclear model. While most of the alpha particles did pass through the foil, some were deflected at large angles, and a few even bounced back. However, to his astonishment, Rutherford observed something unexpected. The prevailing theory at the time suggested that the alpha particles would pass straight through the foil with minimal deflection. In this groundbreaking experiment, Rutherford directed a beam of positively charged alpha particles at a thin piece of gold foil. One of Ernest Rutherford’s most famous experiments is the gold foil experiment. Rutherford’s work continues to have a lasting impact on the field of science. 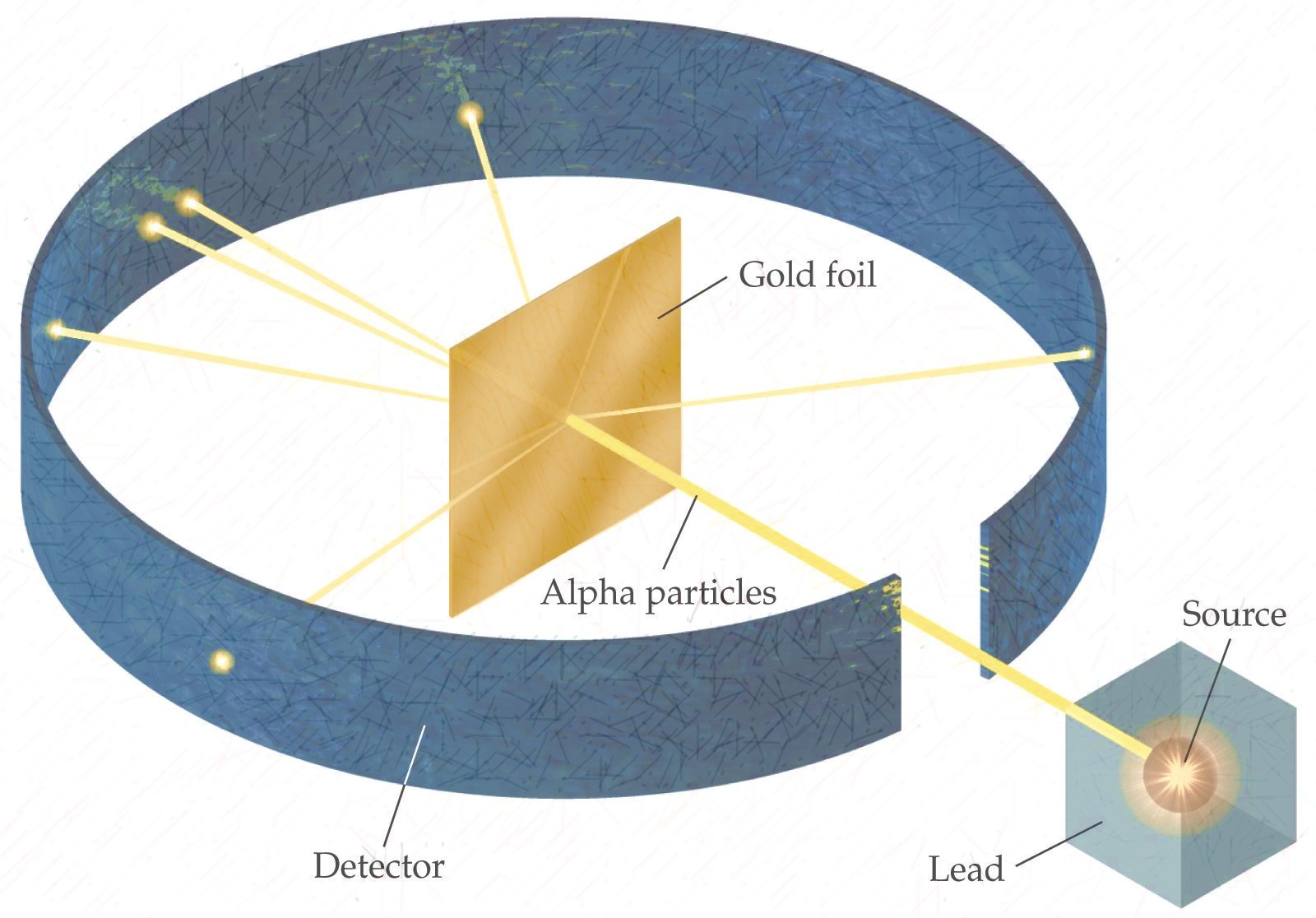
His discoveries in radioactivity led to further advancements in nuclear physics and chemistry.Rutherford’s contributions revolutionized atomic theory and our understanding of the periodic table.The Rutherford model described the atom as having a tiny, dense nucleus surrounded by negatively charged electrons.Rutherford’s experiments on the periodic table showed that atoms are mostly empty space with a dense nucleus.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
